
Infant Brain Damage
Infant brain damage is a serious condition that affects thousands of babies each year. Although there are countless causes, the outcome usually means that the baby experiences long-term permanent neurological problems and, a wide range of physical problems. To better understand infant brain damage, it’s important to educate yourself on how these types of infant brain injuries occur, the symptoms to look out for, how they are diagnosed, and finally, which treatment methods may be best for your child.
What is Infant Brain Injury?
Over 1 million people currently have some form of brain damage in the United States, and an overwhelming amount of these people are infants. Brain damage occurs in many forms and can impact various parts of the body. An infant may experience a variety of disabilities and long-term effects on his or her development, depending on the type of brain damage and the severity of the injury
The medical costs associated with infant brain damage can be astronomical, even reaching millions of dollars each year. Infant brain damage typically occurs during pregnancy, during the delivery process, or shortly after birth. A variety of factors can cause infant brain damage, and it’s important to look out for any signs if you suspect your baby is injured.
Infant brain damage can occur in any part of the brain. However, there are three parts of the brain that are most commonly affected by birth injuries. These are:
Cerebral Cortex
The cerebrum is the uppermost part of the brain that makes up the two hemispheres or the “wrinkly” part of the brain most often seen in illustrations. The hemispheres are connected by the corpus callosum. The cerebrum has many important functions for the brain and body, including:
- emotions and mood.
- judgment.
- movement.
- sensation.
- vision.
- memory.
- language.
Both hemispheres overlap in control of these functions. However, research shows that, in most people, the left side focuses more on language processing and logic. The right side focuses more on processing visual information and creativity.
Cerebellum
The cerebellum is the back portion of the brain. It is primarily responsible for balance, coordination, and some eye movements. The cerebellum does the following:
- Maintains balance.
- Coordinates muscle movements.
- Provides posture control.
Injury to the cerebellum can significantly impact an infant’s ability to move, control posture and muscles and maintain balance.
Brainstem
The brainstem is the middle section of the brain. Its primary function is regulating essential functions throughout the body. The brainstem is like the highway of the body. It helps the cortex and cerebellum communicate with the rest of the body.
The brainstem is responsible for:
- Breathing.
- Swallowing.
- Coughing.
- Sneezing.
- Heart function.
- Vomiting.
It will be important for your healthcare team to determine where the damage occurred and what functions may be affected; in case your infant is suffering from brain damage.
Types of Infant Brain Damage
Birth injuries involving brain damage are among the most serious. They can be difficult to diagnose, and treatment may last for a long period of time. While there are numerous different types of brain damage, there are some that are more common in relation to birth injuries. These are:
- Hypoxic-ischemic encephalopathy (HIE)
- Intracranial Hemorrhage
- Hydrocephalus
- Caput Succedaneum
- Kernicterus
- Traumatic physical injuries
There are numerous possible causes for these birth injuries. There are also many possible treatment options depending on the type of injury your child has, and how severe the damage is. Below, we discuss these factors in more detail.
What Causes Infant Brain Damage
Infant brain damage can result from a variety of situations. In terms of birth injuries, the most common causes of brain damage include the following:
Oxygen Deprivation (Birth Asphyxia)
Oxygen deprivation, also known as birth asphyxia, occurs in about four of every 1,000 full-term births. Premature babies are at higher risk of being injured by oxygen deprivation. Shortly after delivery, infants begin to breathe on their own. Therefore, if they are deprived of oxygen, there is only a small window of time that physicians have in order to treat the problem. Even a few minutes of oxygen deprivation can lead to lifelong brain damage and disorders such as cerebral palsy, autism, attention deficit hyperactivity disorder, impaired vision, and more.
One of the leading causes of infant brain damage is lack of oxygen shortly after birth. Per the American Journal of Neuroradiology, newborn asphyxia happens at an alarming rate: at least two to 10 out of every 1,000 newborns experience a lack of oxygen. Unfortunately, with the correct medical monitoring and treatment, many cases could have been prevented beforehand. It is important to receive regular prenatal checkups during pregnancy to ensure physicians have the best chances of detecting any abnormalities.
Brain damage caused by lack of oxygen falls into two different categories – anoxia and hypoxia.
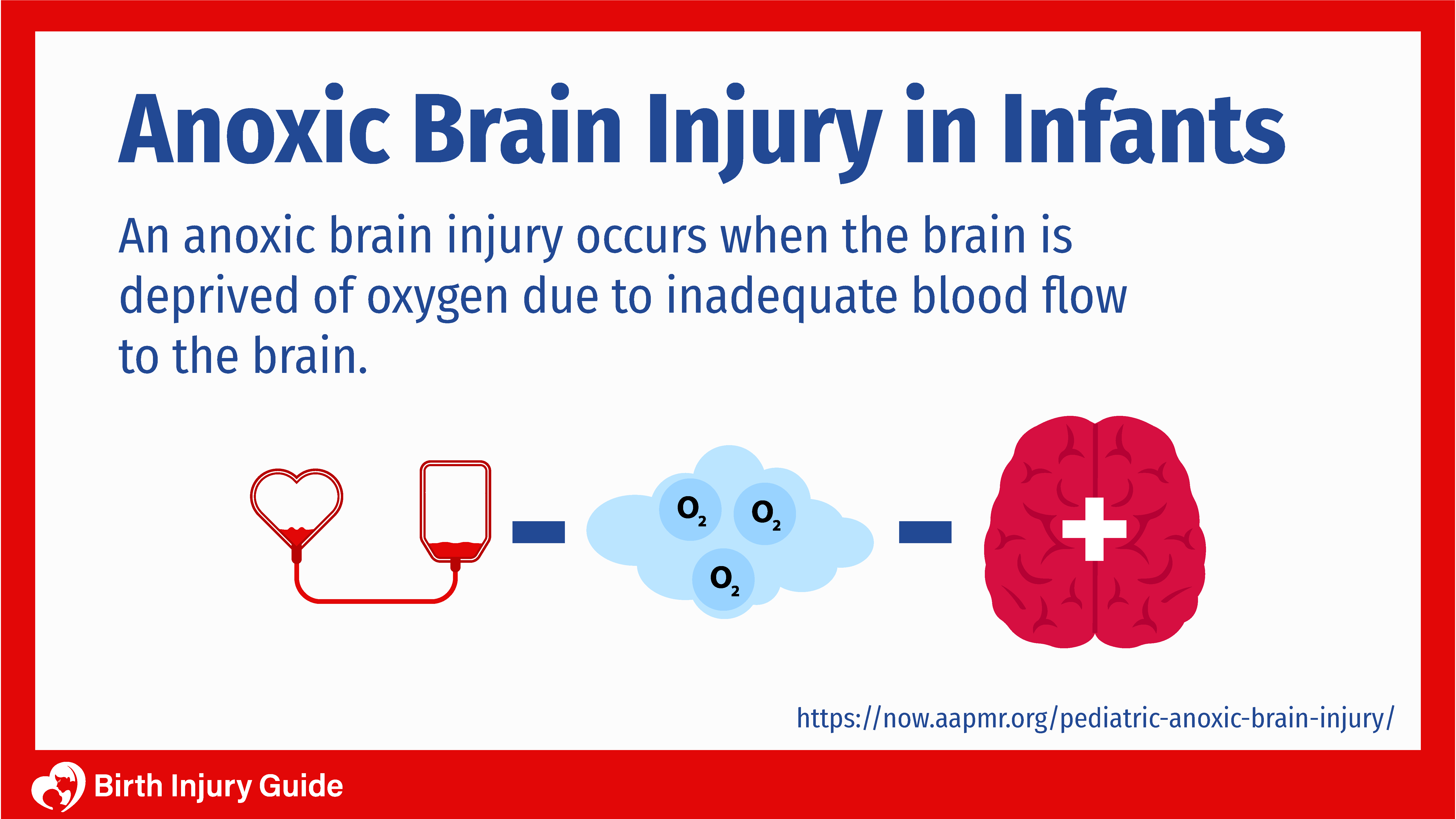
- Anoxia: Occurs when the infant undergoes a total lack of oxygen, which can lead to severe brain injury.
- Hypoxia: Occurs when the infant is deprived of an adequate amount of oxygen. Subsequently, leading to mild to moderate brain damage.
Both anoxia and hypoxia can lead to cerebral palsy and a host of other medical disorders. If the baby is diagnosed with a hypoxic-ischemic injury, it would mean that the baby’s brain was deprived of oxygen and blood.
Most common reasons infants are deprived of adequate oxygen at birth are:
- Umbilical Cord Problems: A twisted, prolapsed, or knotted umbilical cord can cut off an infant’s oxygen supply at the most critical moments of their lives. Furthermore, if an umbilical cord gets wrapped around an infant’s neck, doctors only have a few precious minutes to unwrap it before oxygen deprivation begins.
- Birth Canal Problems: An infant may get stuck in the birth canal if a mother undergoes difficult labor. If medical intervention doesn’t happen quickly, the infant risks losing oxygen. Unfortunately, babies who are lodged in the birth canal are also at risk for other injuries like Erb’s palsy and limb injuries.
- Blocked Airways: Mucous can sometimes become lodged in an infant’s lungs, constricted their airways. Again, healthcare providers must act quickly in order to prevent long-term medical problems.
- Placental Abruption: If the placenta and uterus separate too rapidly, the infant may not get the proper amount of oxygen supply.
Two stages of injury can occur in cases of oxygen deprivation. The first stage involves brain cell damage that occurs within the first few minutes due to a lack of blood flow and oxygen. Reperfusion injury would be the second stage. It occurs after the blood and oxygen flow is restored. Reperfusion injuries may last for periods that range between a few days and several weeks.
Infants with mild or moderate oxygen deprivation often have complete recoveries, especially if they receive prompt medical assistance and physical therapy. Babies whose brain cells were deprived of oxygen for a long period are more likely to suffer permanent damage to their brains, cardiovascular system, respiratory system, digestive organs, and kidneys.
In extreme cases, oxygen deprivation can lead to complete organ failure and death.
Jaundice
Infant jaundice is a common condition, especially in premature babies. It is a yellowish staining of a baby’s skin and eyes. Jaundice is caused by an excessive amount of bilirubin, which is a yellow pigment in red blood cells, in the baby’s blood.
If infant jaundice is treated immediately, there usually isn’t a reason for concern. However, if left untreated, bilirubin levels in the baby’s circulatory system increase. These higher levels of bilirubin cause a condition called kernicterus. Kernicterus occurs when excess bilirubin levels cross the blood-brain barrier and damage the brain.
Symptoms of kernicterus may vary, but may include:
- Lethargy/drowsiness
- Poor feeding
- Fever
- High-pitched crying
- Absence of certain reflexes
Kernicterus falls into three different stages, with the earliest stage marked by extreme jaundice, lethargy, and difficulty in swallowing and sucking. If it progresses, the infant may experience seizures, high-pitched wails, and a protruded fontanel (also known as a baby’s “soft spot” on the top of the head). The final stage of kernicterus is marked by intellectual loss, hearing loss, seizures, movement disorders, and rigid muscles. In some extreme cases, infants may die from kernicterus. Treatment must start immediately and typically entails blood transfusions and light therapy.
Physical Trauma
According to the University of California, San Francisco (UCSF) School of Medicine, birth related brain injuries are the leading cause of cerebral palsy and intellectual disabilities in the United States. Babies are most at risk of suffering brain injuries during the birth process, especially if they are premature.
During labor and delivery, a host of medical mistakes and other forms of physical harm may lead to infant brain damage. Injuries like brachial plexus injuries are more common physical injuries than brain injuries. However, physical trauma during labor and delivery can lead to brain damage. Some examples include:
- Excessive Pulling: During delivery, if a doctor pulls too hard or in an abnormal way, a host of injuries may occur, including brain damage.
- Abnormal Use of Birth-Assisting Tools: Tools such as vacuum-extraction or forceps must be used with experience and caution when delivering an infant. If used incorrectly or with too much force, brain damage or other physical injuries may occur, such as a hematoma on the baby’s head.
- Difficulties During Labor: Extended periods of pushing and contractions during a long, difficult labor can result in brain damage. This typically occurs when the baby’s brain and head are constantly knocked and pushed against the mother’s pelvic bones.
Maternal Infections
Although maternal infections are a relatively common complication during birth, they are also another major cause of the onset of infant brain damage. Brain damage due to maternal infections may occur during pregnancy, or during the delivery process. If a physician monitors, detects, and treats any infections, the chance of infant brain damage is significantly reduced.
The most common types of maternal infections that lead to infant brain damage include:
Intrauterine Infections
Intrauterine infections happen while the infant is still in the womb, and can lead to brain damage before the infant is born. The severity of risks will depend on the type of infection as well as how far along the pregnancy is. Examples of intrauterine infections include rubella, herpes, and cytomegalovirus. The infections are most dangerous during the first trimester of pregnancy.
Rubella
Rubella is considered to have been ‘eliminated’ in the United States thanks to vaccines. However, the infection can occur if someone who is not vaccinated is exposed to someone who is infected. If a woman who has rubella gets pregnant, she can transmit the infection to her child. Rubella is extremely damaging to a developing fetus, especially during the first trimester. Infants exposed to rubella are at risk for birth defects, heart defects, brain damage, and more.
Chickenpox
Varicella, or chickenpox, is a virus that causes a painful, itchy rash all over the body. It is highly contagious. Infants can develop chickenpox from their mother if she is infected during the last weeks of her pregnancy. Chickenpox is considered a severe illness in newborns and can cause pneumonia, inflammation of the liver, and inflammation of the brain.
Group B Strep
Group B streptococcus (GBS) is a bacteria that is common in the genital tract and gut. Most often, it is dormant and the individual may experience no symptoms. However, GBS can be transmitted to the infant during pregnancy, labor, and delivery. An infant GBS infection can cause severe illness and brain damage. In the most severe cases, it can be fatal.
Meningitis
Meningitis occurs when the membranes around the spinal cord and brain become inflamed. This is in response to an infection, such as GBS or E. coli. Inflammation of the brain caused by meningitis can lead to permanent brain damage if not treated accordingly.
In a Danish clinical study, babies of women with maternal infections can also be at higher risk of developing childhood epilepsy, cognitive issues, and schizophrenia. Maternal infections may also lead to premature birth, which in turn heightens the risk of infant brain damage and cerebral palsy. Some estimates suggest that 30-40 percent of all premature births in the United States may be the result of intrauterine infections.
Dropped Glucose Levels
Infant hypoglycemia, or low glucose levels, occurs most frequently in premature infants, infants who have a low birth weight, or infants who are large for their gestational age. Infant hypoglycemia can also occur if the mother has diabetes. Glucose levels must always be checked after delivery, especially if the mother experiences an arduous, tiring labor and delivery process. If glucose levels are found to be too low, medical intervention is required immediately. Waiting too long may result in brain damage.
Can Infant Brain Damage be Prevented?
Preventing infant brain damage begins during pregnancy. Proper prenatal care is essential to diagnosing and treating any condition, complication, or infection that could cause problems as pregnancy progresses into eventual delivery. If doctors note a risk factor during pregnancy that could cause infant brain damage, they should label the pregnancy as ‘high-risk’. The mother and her developing fetus should be treated and monitored accordingly.
During labor and delivery, it is important that healthcare providers appropriately monitor mother and infant. Any signs of fetal distress should be taken seriously and should be immediately acted upon. That may mean more consistent monitoring of fetal vital signs, or ordering a c-section to prevent oxygen deprivation.
During and after delivery, it is also important that healthcare providers take measures to prevent head trauma. During delivery, head trauma may occur as a result of pressure, use of delivery assistance tools, or a doctor who uses too much force. After delivery, healthcare providers must monitor the infant for signs of fetal distress, jaundice, stroke, or infection. They must also be careful to properly handle and support the infant’s head to prevent injuries.
Failure to properly care for an infant before, during or after delivery is medical negligence.
Start Your FREE Case Review Today
If you or your child is injured as a result of medical negligence, call us to learn more.
Symptoms of Infant Brain Damage
Brain damage can happen either from blunt force trauma to the infant’s head (less common, but possible with a forceps extraction injury), from a long labor where the brain is compressed in the birth canal for far too long, or, more likely, from a difficult labor that at one point results in the infant losing oxygen.
Some of the first signs and symptoms of brain damage can be detected shortly after birth by looking at the infant’s appearance. Although not always present in all infants who suffer from brain damage, some of the common infant brain damage symptoms shortly after birth include:
- Abnormally large forehead
- Abnormally-shaped spine
- Distorted facial features
- Unusually small head (more prominent in smaller infants)
- Seizures
- Neck stiffness
- Difficulties in focusing the eyes
Infants with brain damage may also exhibit an abnormal temperament, with symptoms including:
- Troubles with sleeping while lying down
- Excessive crying
- Problems with feeding
- Excessively fussy for no apparent reason
Developmental Delays
Some signs of infant brain damage may not be evident until the child starts missing developmental milestones. These symptoms of brain damage are calculated in a few different ways: cognitive, perceptual, physical, and behavioral or emotional. They may be considered developmental delays, or a condition further classified as a developmental disability.
Cognitive symptoms
Cognitive symptoms are usually identified as the child grows older and possibly misses normal developmental steps. The most common symptoms include difficulties with
- Attention and concentration
- Memory and processing information
- Processing language
- Controlling impulses
- Communication
Sometimes brain damage isn’t obvious until children enter school and a learning disability is suspected or diagnosed. Talk to your child’s doctor if you suspect infant head injury symptoms as they get older.
Perceptual Symptoms
Perceptual symptoms are sometimes more difficult to diagnose, but symptoms include:
- Spatial disorientation
- Changes in vision and/or hearing
- Heightened sensitivity to pain
Physical Symptoms
As the infant grows older, additional physical symptoms may manifest. Some physical symptoms may be difficult to diagnose (such as headaches) whereas other symptoms may be more obvious. Some physical symptoms include missing developmental milestones such as:
- Crawling
- Pulling themselves up without assistance
- Walking
- Running
- Hopping or skipping
- Walking up and down stairs without assistance
- Feeding themselves without assistance
- Tying shoes, drawing, and coloring without assistance
- Sitting up alone, without assistance (learn more about when babies start sitting up on their own)
- Getting dressed without assistance
- Holding things without dropping them and with a firm grasp
Some of the symptoms not related to milestones include:
- Light sensitivity
- Paralysis
- Tremors
How Brain Damage Affects Children
Since brain injuries are so complex and no two injuries are the same, how the injury affects children will vary. The good news is that most infant brain injuries are mild, and in many instances, infants are able to fully physically recover with the right treatment. It may take a long time for infant brain damage recovery, sometimes even years, despite myths that people with mild brain injuries recover quickly.
In addition, the baby may have good days and bad days. For example, one day the infant may coo and smile and the next day become inexplicably fussy. This is normal behavior for infants who suffer from mild brain damage, but is also common among infants without brain damage. It can be incredibly frustrating, however, for you and your child.
Since infants and children’s brains are not fully developed yet, even mild brain damage may result in permanent cognitive and emotional disabilities. These impairments may not be obvious right away and may take a few years to develop. Sometimes the disabilities aren’t discovered until the child enters school.
After a severe brain injury, an infant’s brain begins to change, leading to heightened pressure in the skull. If not treated immediately, it may lead to additional brain damage and possibly death. Surgery and medication are often used to treat severe brain damage. However, the outcome depends upon how quickly the injury was treated and how well the patient reacts to treatment.
Infants and children with brain damage may be affected in the following ways:
Infant Seizures
Seizures occur in one out of every 10 people who experience traumatic brain injuries. Seizures are caused by abnormal signals in the brains. Infants and children may experience seizures for a few days, or, in some cases, they can last for years. Infants and children may develop epilepsy if they have more than one seizure, which can last a lifetime.
Infant Spasticity Issues
When the brain is severely damaged, infants are at risk for spasticity disorders like cerebral palsy. Spasticity affects the way a person crawls, walks, holds objects and completes other daily activities that require major muscle use. Although medication and therapy will help reduce muscle stiffness, contractures and other effects of spasticity, it is often a permanent disorder.
Cognition Issues
Cognitive issues are one of the most prominent consequences of brain damage, and can affect any infant who suffers from brain damage, whether mild or severe. Cognitive problems are usually long-term and marked by memory loss, impaired thinking skills, behavioral problems, and mental health issues such as depression, aggression, anxiety and inappropriate behavior in social situations.
Tests to Confirm Infant Brain Damage
The majority of infant brain damage cases are diagnosed by observing symptoms and conducting tests. Computed tomography (CT) scans and magnetic resonance imaging (MRI) are two tests that provide images of the brain. These tests communicate to the doctor if there is a brain injury, such as a hemorrhage or a skull fracture. In addition, an electroencephalogram (EEG) may also be done. An EEG is a test that calculates the electric pathways of the brain to make sure that communications are normal and strong.
It is more difficult to assess the brain and mental health of infants and children under two years of age. An MRI and head ultrasound can be helpful in assessing brain health and any possible brain damage.
Infant Brain Damage Treatment
Treatment of infant brain damage will depend upon the type and seriousness of the injury. If your infant suffers from brain damage, it is important to know what treatment options are available so that you and your child’s healthcare team can decide what works best for you and your baby. The following treatment options are to be used only as a guideline for the most common treatments of infants and children with brain damage. Common treatment options include:
Medication
Many infants who suffer brain damage experience seizures, head pain, swelling and infections. Physicians usually monitor the baby closely to determine what type of medications would work best to help relieve these symptoms. Some of the medications used for brain injuries include:
- Anti-Seizure Medications: Anti-seizure medications, such as phenytoin, benzodiazepines or levetiracetam, helps control spastic movement and seizures.
- Behavioral Medications: Behavioral and concentration medications are typically prescribed when the infant grows older and exhibits common side effects of brain damage, including hyperactivity, attention deficits disorders (ADD or ADHD) or aggression. Common medications include bromocriptine and methylphenidate.
- Diuretics: Diuretics promote an increase in urination, which in turn helps reduce the fluids that build up in bodily tissues, including those around the brain. Diuretics are administered intravenously in the acute setting to reduce initial brain swelling. The most common medication for patients with brain damage is Mannitol.
Therapy
Infants with brain damage are likely to develop emotional, cognitive and physical disabilities. According to the British Medical Journal (BMJ), disorders including ADD, ADHD, autism, learning disorders and behavioral problems have a strong correlation with newborn brain injuries.
In addition, infants with severe brain injuries may need lifelong assistance with physical development, including walking, balance and coordination, standing, sitting down, and daily tasks such as eating, drinking, and feeding themselves. This is why therapy is so important for infants and children with brain damage. Therapy can help the child develop and become more independent.
Physical Therapy
Physical therapy is one of the most common types of treatment for birth injuries. During therapy, physical therapists work with patients to improve and help develop more physical independence by teaching and monitoring a series of exercise and training, which may include:
- Flexibility exercises
- Coordination and balance exercises
- Strength training
- Balance activities that help children learn to sit and stand successfully
- Thermal treatments and electrical stimulation
- Joint mobilization activities
It’s important to note that there are various physical therapists who offer different treatment options and rehabilitation plans. When choosing a physical therapist for your child, make sure that they have experience working with children who suffer from brain damage. Furthermore, the physical therapist should be board-certified as a clinical specialist. Ideally, you should also look for a physical therapist who is a Certified Brain Injury Specialist (CBIS).
Occupational Therapy
Per the Brain Injury Association of America (BIA), occupational therapy helps people overcome challenges that prevent them carrying out daily living tasks. For instance, children with brain damage may need help with:
- Using the toilet alone
- Feeding and swallowing
- Grooming and dressing
- Cognitive issues
- Emotional issues
Occupational therapists assess individual problems and then create a plan that will help the patient learn the skills they are lacking in. For instance, infants who need help with feeding may get assistance via a feeding tube until they are old enough to transition to oral feeding. An occupational therapist will help teach the child how to eat without a feeding tube through a series of exercises and activities.
Another example includes a child with autism or an attention deficit disorder; the therapist will create an individualized plan to help the patient deal with cognitive and emotional challenges. Other occupational therapy treatment includes:
- Neuro-musculoskeletal activities
- Rehabilitation for fine motor skills
- Sensory and perceptual processing
- Oral motor skills rehabilitation
Therapeutic Hypothermia Treatment
Therapeutic neonatal hypothermia, or cooling therapy, reduces an infant’s body temperature in order to stop brain damage and prevent additional brain damage. Cooling therapy has shown positive results if administered immediately after birth.
Cooling therapy works by cooling the infant’s body temperature to around 89°F to 93°F or 32-34 C. This temperature is maintained for at least three days. According to the National Institutes of Health (NIH), cooling therapy not only increases an infant’s chance of survival, but also decreases the chances of long-term permanent brain damage and physical impairment.
Surgery
Surgery is most often used for infants who have experienced severe head trauma during the delivery period. For instance, if the birth injury caused bleeding in the skull or brain, or if there are skull fractures, surgery may be an option. Surgery can often repair the fractures, stop the bleeding and prevent more damage to the vessels. It is important to resolve an infant brain bleed as quickly as possible.
Infant Brain Damage Prognosis
The prognosis for brain damage depends on the severity of the injury and other health-related factors. If healthcare providers identify a brain injury and start treatment immediately, infants may survive the injury with a positive prognosis. According to the Simon’s Foundation Autism Research Initiative (SFARI), infants who experience brain injuries are at high risk for developing:
- Autism
- Attention deficit disorders
- Emotional outbursts
- Physical disabilities
These conditions can impact various aspects of daily life, and may require ongoing care throughout the child’s life. However, children with these disabilities can go on to live productive lives. This is especially true of children who have access to physical and occupational therapy soon after their injury.
If the injury is severe, or is not immediately diagnosed, then there is a greater chance that the child will suffer more serious consequences, such as:
- Cerebral palsy
- Seizures
- Physical delays
- Developmental delays
These conditions are often permanent, and your child may require a lifetime of specialized care. Even so, with the right treatment, therapy plan and support, your child can have a happy and fulfilling life.
How to Optimize Your Child’s Prognosis
As a parent, you have a key role in helping your child heal and move past his or her brain injury. Together with your child’s healthcare team, you will provide support for your child at home and school. Of course, you want what is best for your child, and you want them to have every opportunity to thrive.
There are some things that you can do to optimize your child’s prognosis, such as:
- Talk to your child’s doctors about things you can do at home to help your child.
- Work with therapists and create a list of activities that you can do at home with your child to support what they are learning during therapy sessions.
- Consider family counseling with a psychologist or psychiatrist who is experienced with brain injury cases. This can help your entire family better understand the changes that may happen in your home.
- Encourage your child to set small, reasonable goals. Praise him or her when those goals are met. Be supportive and encouraging when they are not.
- Develop a lifestyle routine for your home and family that is as “normal” as possible.
- Reach out to your community for support. There are often state and local networks dedicated to families suffering from certain types of injury or illness.
- When it’s time for school, work with your child’s teachers to develop a plan for academic success. Your child may need special accommodations or an individualized education plan (IEP).
Of course, parenting a child with a brain injury or a long-term condition like cerebral palsy can be difficult. It is important to remember that you are not alone. There are various options to find support and even financial assistance if you need it. If you believe your child’s brain injury is the result of medical negligence during birth, legal guidance is also available.



